סמינר בכימיה אורגנית
Dr. Doron Pappo, Ben Gurion University
Iron-Based Oxidative Cross Coupling of Phenols: Method Development, Mechanism and Applications.
Abstract:
Single electron oxidative coupling of phenols, catalyzed by metalloenzymes, is a common synthetic tool used by plants for the rapid construction of a variety of phenolic natural products, necessary for the plants’ growth and protection. Despite extensive studies to elaborate similar tactics in the lab, most of these methods were limited to dimerization and oligomerization of phenolic building blocks and to intramolecular transformations.
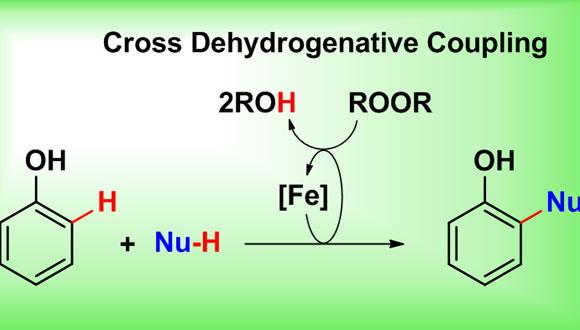
While most of the studies in metal catalyzed cross coupling reactions have focused on coupling between nucleophiles and electrophiles, new developments in metal catalyzed oxidative cross coupling reactions enable the cross coupling of two nucleophiles. The identification of specific conditions for the activation of two C-H bonds by utilizing earth-abundant iron and copper metals in the presence of complementary oxidant is of great interest for the development of sustainable and economical chemistry.
In this talk, I will describe our group’s efforts in developing novel biomimetic iron-based oxidative cross coupling reactions between phenols and various C-H nucleophiles, such as 1,3-dicarbonyls and conjugated alkenes. These simple, yet highly efficient, methods provide direct entry to complex phenolic bioactive natural products in a step- and atom- economical manner. The two-step total synthesis of coumestrol and the results from our preliminary mechanistic study will be discussed as well.